Stricter rules needed on drug approvals
Updated: 2013-01-29 13:34
(China Daily)
|
||||||||
China needs more stringent requirements for the approval of generic versions of biologics, complex drugs made from living cells, to ensure their safety and effectiveness, according to an industry report.
Generic copies of biological drugs can be registered as new products in China with fewer testing requirements than originals, according to the report, led by the R&D-based Pharmaceutical Association Committee, which represents drugmaking giants including Roche Holding AG and Pfizer Inc.
China Daily - Agencies

 Li Na on Time cover, makes influential 100 list
Li Na on Time cover, makes influential 100 list
 FBI releases photos of 2 Boston bombings suspects
FBI releases photos of 2 Boston bombings suspects
 World's wackiest hairstyles
World's wackiest hairstyles
 Sandstorms strike Northwest China
Sandstorms strike Northwest China
 Never-seen photos of Madonna on display
Never-seen photos of Madonna on display
 H7N9 outbreak linked to waterfowl migration
H7N9 outbreak linked to waterfowl migration
 Dozens feared dead in Texas plant blast
Dozens feared dead in Texas plant blast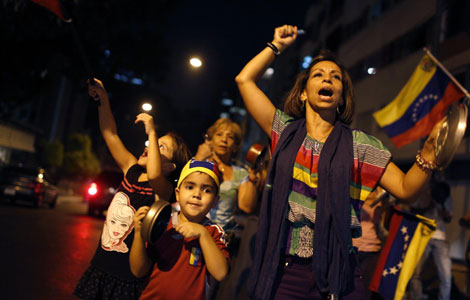
 Venezuelan court rules out manual votes counting
Venezuelan court rules out manual votes counting
Most Viewed
Editor's Picks

|

|

|

|
|

|
Today's Top News
Boston bombing suspect reported cornered on boat
7.0-magnitude quake hits Sichuan
Cross-talk artist helps to spread the word
'Green' awareness levels drop in Beijing
Palace Museum spruces up
First couple on Time's list of most influential
H7N9 flu transmission studied
Trading channels 'need to broaden'
US Weekly

|

|